Directive on Patients' Rights in Cross-border Healthcare: Press release, Questions and answers, Informative video and leaflet
EU citizens have the right to access healthcare in any EU country and to be reimbursed for care abroad by their home country.
Directive 2011/24/EU on patients’ rights in cross-border healthcare sets out the conditions under which a patient may travel to another EU country to receive medical care and reimbursement. It covers healthcare costs, as well as the prescription and delivery of medications and medical devices.
Key elements
With health policies and systems increasingly interconnected, the Directive makes it easier to access:
- Information on available healthcare in other European countries
- Alternative healthcare options, and/or specialised treatment abroad.
The provisions strike the right balance between maintaining the sustainability of health systems, while protecting patients' right to seek treatment outside their home country.
The Directive:
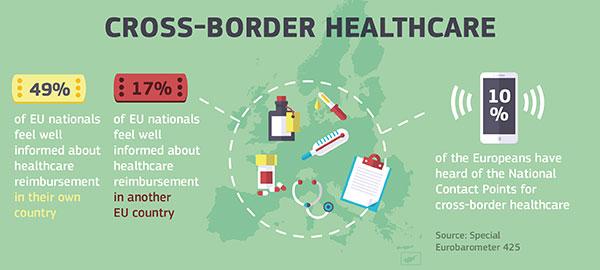
- Creates a network of National Contact Points to provide clear, accurate information on cross-border healthcare
- Creates EU rules on a minimum list of elements to be included in a medical prescription taken from one EU country to another (cross-border prescription)
- Encourages further development of European Reference Networks of medical expertise, broadening cooperation between EU countries, with added benefits to health technology assessments and e-health.
Monitoring the Directive
- A 2018 European Commission report (available in all EU languages) and its annex covers the key provisions, trends and progress to date in the last reporting period (2015-2018).
- Data report on the application of the Directive in EU countries. (Year 2015, 2016, 2017, 2018, 2019)
- A 2015 European Commission report (available in all EU languages) set out the state of play, and covers the most important and relevant provisions.
Cross-border Healthcare Expert Group
The Cross-border Healthcare Expert Group brings together healthcare representatives from all EU countries to assist the European Commission with the implementation of the Directive. It provides the Commission with advice and expertise, and national authorities with a forum to exchange their experiences of the Directive.
To know before you go
- The terms and conditions which apply both to the healthcare you will receive, and any reimbursement to which you are entitled
- Your rights in accessing healthcare in another EU country
- The National Contact Point in your country
- The quality and safety of the treatment
- Complaints procedures in case something goes wrong
Related information
- 'To know before you go'
- Your questions on cross-border healthcare
- Practical information on healthcare in another EU country (Your Europe)
- National Contact Points
- Video:

Details
- Publication date
- 25 October 2013
- Author
- Directorate-General for Health and Food Safety
